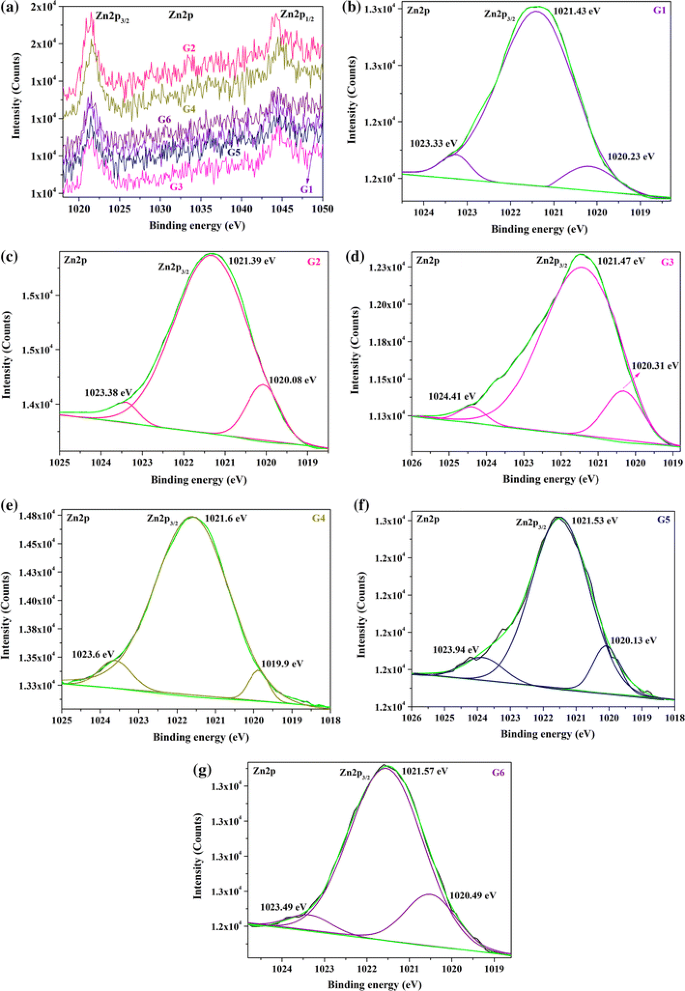
X-ray photoelectron spectroscopy is a powerful technique for the chemical analysis of the surface of materials. Inset tables in rows c and d summarize the calculated atomic ratios relative to the total amount of copper present. C 1s and Na 1s data are normalized by the integrated areas of the corresponding Cu 2p scans.
This webinar covers the basics of X-ray photoelectron spectroscopy, with a special emphasis on how it can be used in the field of polymer surface analysis. XPS Cu 2p (row a), O 1s (row b), C 1s (row c) and Na 1s (row d) spectra of Cu 2 O films AD after 0 s, 30 s, and 450 s ion sputtering. We present how a dual mode ion source can be used for multi-technique sample analysis, showing the importance of sample cleaning and how depth profiles can be performed. The drawing shows the relation between the Silicon atoms and their XPS peaks. Here is an example of Si (2p) spectrum from a Synchrotron facility. Multi-technique Surface Analysis and Cluster Ion Sample Cleaning Silicon peak-fitting is can be very challenging. XPS delivers chemical state information from the topmost nanometers of a sample surface, enabling you to measure passivation coatings, understand catalyst chemistries, and develop bio-compatibility coatings. However, all elements having a minimum concentration of ca.
#Na xps peak software#
This webinar is designed to offer training to current users of Avantage software and act as an introduction to those unfamiliar with it.Įxplore a number of applications for thin film coating analysis including forensic studies, graphene, multi-layered glass, coated fabrics and photovoltaics. Peak C 1s 0.278 F 1s 1.000 O 1s 0.549 O 2s 0.033 Ag 3d 5/2 3.592 SF Different elements have a different sensitivity and will be analyzed with different minimum concentration limits. Understanding Surface Chemistry with Avantage Software
#Na xps peak how to#
From the measured n/p junction depths, an estimate of the diffusion coefficient was made in the range of 10-10 to 10-8 cm2/sec for sodium in multi-crystalline CuInSe2 at 250 ☌.Understanding XPS images and depth profiles with Avantage Software - Part 2īy watching the webinar, you will learn how to use the right tools to understand multi-level data sets, such as depth profiles and images. As a consequence, the resulting chalcopyrite becomes stoichiometrically selenium deficient and hence n-type. The 59 eV peak has been assigned to Na2SeO3, created by oxidation of Na2Se, itself formed by the combination of sodium with selenium destined for the creation of the CuInSe2. This peak was also seen earlier in Bridgman CuInSe2, where Na was introduced into the melt prior to compound synthesis, for ingots stored for a long time in air. 2(C) and (D) 0/black curves at the bottom) the well-known signature of TCNQ including the shake-up peak approximately 2.6 eV at higher binding energies in relation to the core level peaks (399.3 eV for N1s) for the N1s and C1s core levels.During the first three deposition steps of sodium onto TCNQ a sodium peak appears at 1071.6 eV and shifts. As a result, a single XPS Se 3d peak was observed at 54.5 eV but, after subsequent annealing the same samples in air in a sealed ampoule at 200 ☌ for 1 hour, a second Se 3d peak appeared at a binding energy of 59 eV. The XPS analysis of the pristine film shows (Fig.
The amount of Na diffused in was monitored by the intensity of the XPS Na 1s peak at a binding energy of 1,071 eV.

Using stoichiometric p-type samples of Bridgman-grown CuInSe2, elemental sodium was diffused into the surface, within an ampoule under vacuum, causing conversion of the samples to n-type to a depth of more than 100 microns from the surface. However, in some papers, when checking the peak shift of XPS, it is said that CVD MoS2 is more p-type doping because CVD MoS2 has a lower shift of XPS peak than exfoliated MoS2.
